Pain Management Supplies Built for Procedure-Day Reliability
Credentialed access to interventional pain injectables and procedure supplies used in office-based and ASC procedures.
Pipeline Medical provides credentialed access to injectables and interventional supplies used in office-based and ASC pain procedures. All products are sourced through authorized manufacturers and evaluated for sterility, compliance, and procedural reliability.
Designed for clinical environments, our structured sourcing model helps pain practices streamline inventory management, reduce vendor fragmentation, and maintain consistency across procedural schedules.
- Invoice Consolidation
- Easy Reordering For Repeat Procedures
- Authorized Sourcing With Lot Traceability
- No Hidden Fees
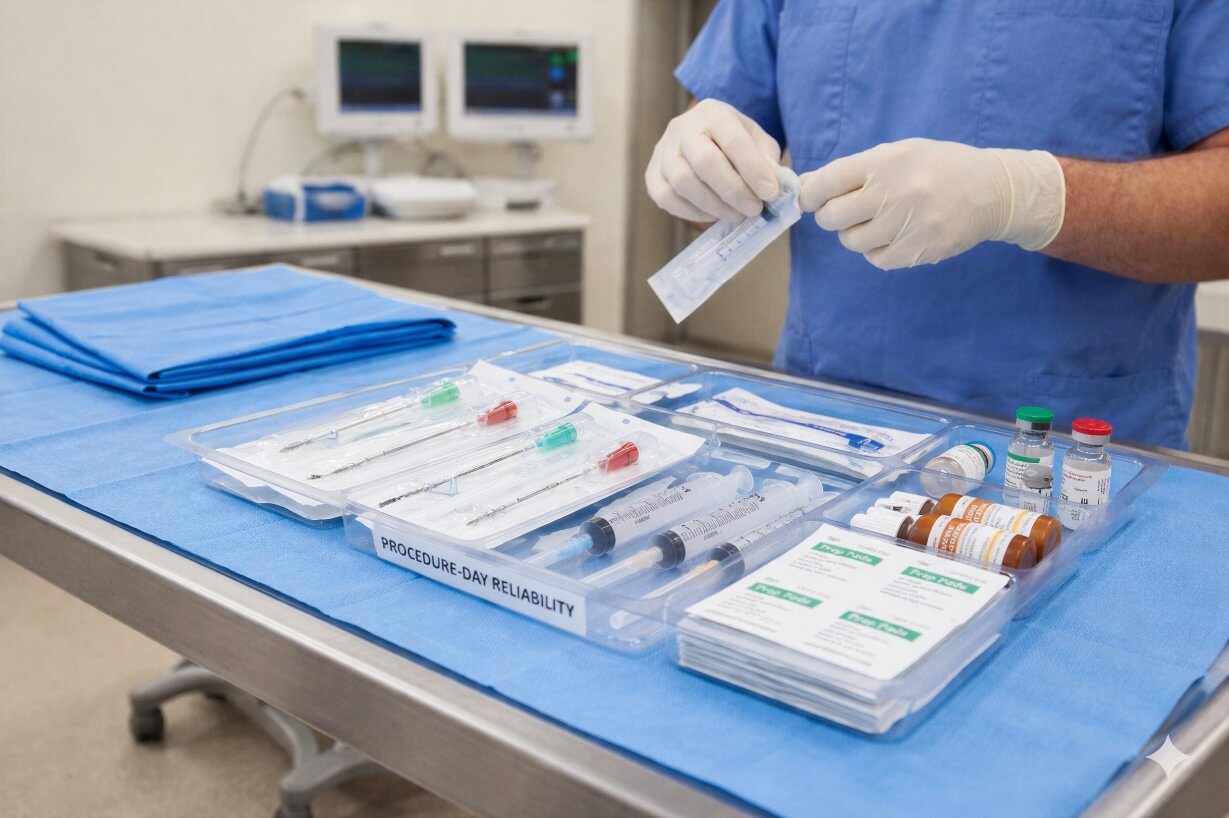
Why Pain Management Practices Trust Pipeline Medical
Reduce vendor sprawl, simplify invoicing, and keep procedure days on schedule.
Centralized Supply Access
Aligned with procedural care. Get centralized access to essential injectables and procedure supplies used across interventional pain workflows.
Verified Sterile Products
Focused on sterility and clinical use. Access pharmaceuticals and consumables from authorized manufacturers and evaluated for procedural reliability.
Ordering Built for Procedure Volume
For high-frequency pain management schedules. Our sourcing model supports predictable reordering, volume alignment, and inventory consistency.
Workflow-Aware Account Support
Support informed by procedural environments. Our account managers understand the supply dynamics of nerve blocks, epidural injections, and other procedures.
Procedure-Day Supply Checklist
Interventional pain procedures depend on consistent access to sterile consumables and injectables.
The checklist below reflects typical components used in regional anesthesia and pain procedure trays.
Prep Components
| Description | Size |
|---|---|
| Povidone-lodine Solition | 1 oz. |
| Sponge Sticks | - |
| Orange Chloraprep | 3 mL |
Procedural Needles
| Description | Size |
|---|---|
| 18g × 3.5" Tuohy Epidural | |
| 22g x 3.5" Quincke Spinal | |
| 17g × 3.5" Tuohy Epidural | |
| 18g × 3.5" Hustead Epidural | |
| 20g x 3.5" Tuohy Epidural | |
| 22g × 3.5" Tuohy Epidural | |
| 18g × 3.5" Quincke Spinal | |
| 20g × 3.5" Quincke Spinal | |
| 22g× 5" Quincke Spinal | |
| 22g×7" Quincke Spinal | |
| 25g × 3.5" Quincke Spinal | |
| 25g × 3.5" Whitacre Spinal | |
| Other |

Hypodermic Needles
| Description | Size |
|---|---|
| 18g × 1.5" | |
| 22g × 1.5" | |
| 25g ×1.5' | |
| 25g × 5/8" | |
| 27g × 1.25" | |
| 27g × 1.5' |

Syringes
| Description | Size |
|---|---|
| 3mLL/L | |
| 5mL L/L | |
| 10mL L/L | |
| 3mLL/S | |
| 5mL L/S | |
| 10mL L/S | |
| 20mL L/L | |
| 20mL L/S |

LOR Syringes
| Description | Material |
|---|---|
| 5mL LOR L/L | Glass |
| 5mL LOR L/S | Glass |
| 7mL LOR L/L | Plastic |
| 7mL LOR L/S | Plastic |
| 10mL LOR L/L | Glass |
| 10mL LOR L/S | Glass |

Drapes and Towels
| Description | Material |
|---|---|
| Fenestrated Drape (3" x 4" opening) | Paper |
| Fenestrated Drape (4" opening) | Plastic |
| Fenestrated Drape (4" x 8" opening) | Plastic |
| Absorbent Towel | - |
| OR Towel | - |
| Other |

Other Components
| Description | Material |
|---|---|
| Filter Needle | |
| Foam Sharps Pad with Adhesive | |
| Drug Label Sheet | |
| Gloves | Size: | |
| Filter Straw | |
| 6" Extension Set Male Luer Lock | |
| 12" Extension Set Male Luer Lock | |
| Skin Marker | |
| Gauze (4x 4 ply) | |
| Other |
Pharmaceuticals
| Description | Size |
|---|---|
| Lidocaine 1% | 5mL |
| Sodium Chloride. 9% | 10mL |
| Lidocaine 1.5% with Epinephrine | 5mL |
| Bupivacaine 0.75% Dextrose 8.25% | 2mL |
Needles and Procedural Access
| Description | Size |
|---|---|
| Epidural needles (e.g., Tuohy, Hustead) | |
| Spinal needles (e.g., Quincke, Whitacre) | |
| Standard hypodermic needles in multiple gauges and lengths |

Sterile Prep and Draping Supplies
| Description | Size |
|---|---|
| Povidone-iodine or chlorhexidine prep solution | |
| Sponge sticks or applicators | |
| Sterile fenestrated drapes (paper or plastic) | |
| Absorbent towels and OR towels |

Syringes and Loss-of-Resistance Equipment
| Description | Size |
|---|---|
| LOR (loss-of-resistance) syringes in glass or plastic | |
| Standard Luer-lock and Luer-slip syringes (3 mL–20 mL) |
Medications and Injectables
| Description | Size |
|---|---|
| Lidocaine | |
| Lidocaine with epinephrine | |
| Bupivacaine with dextrose | |
| Sterile saline (sodium chloride) |

IV / Injection Accessories
| Description | Size |
|---|---|
| Extension tubing sets with Luer lock connections | |
| Filter needles and filter straws |

General Procedure Consumables
| Description | Size |
|---|---|
| Sterile gloves | |
| Gauze (4 × 4) | |
| Skin markers | |
| Drug labels | |
| Adhesive sharps pads or sharps safety accessories |
Our Highest-Demand Interventional Pain Supplies & Pharmaceutical Categories
Structured access to injectables, pharmaceuticals, and procedure-ready supplies used across outpatient and office-based pain settings.
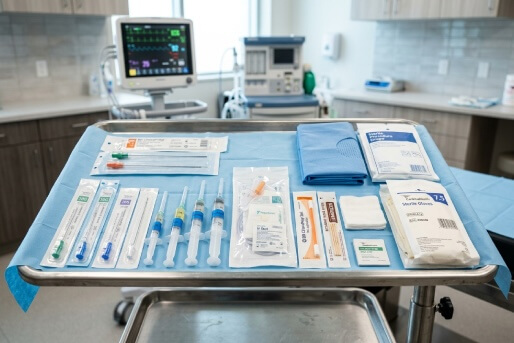
Interventional Pain Procedure Consumables
Sterile, procedure-ready consumables for pain interventions. Includes spinal and epidural needles, syringes, IV start kits, skin prep, drapes, sharps containers, and gloves designed to support efficient turnover in outpatient and office-based pain settings.
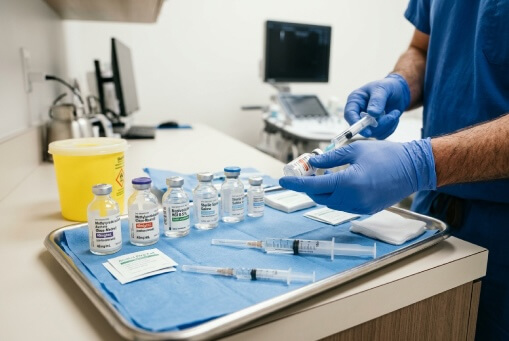
Pain Management Injectables & Medications
Injectable anesthetics, corticosteroids, and contrast agents used in nerve blocks, epidural injections, and facet procedures. Reliable sourcing supports consistent care across pain management workflows.
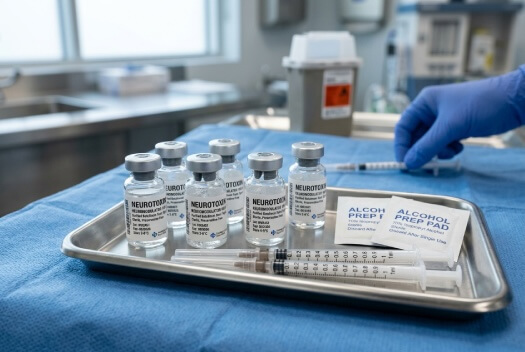
Therapeutic Neuromodulator Injectables
Therapeutic neuromodulator injectables used in clinically appropriate indications. We support compliant ordering and consistent availability in pain practices.
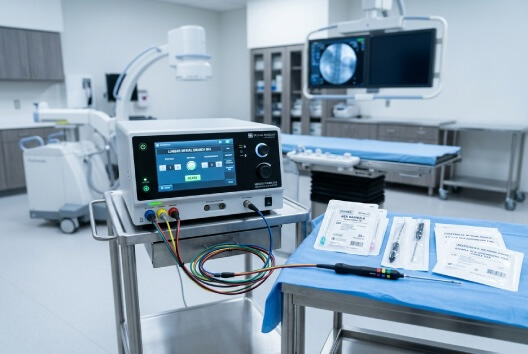
Radiofrequency & Energy-Based Pain Procedure Supplies
Energy-based consumables for radiofrequency ablation (RFA) and lesioning procedures, designed for precision thermal targeting.
Shop Supplies by Procedure
Interventional Spine Procedures
(Epidural Steroid Injection, Medial Branch Block, & Facet Joint Injection)
Radiofrequency Ablation (RFA)
- RF cannulas and RF electrodes
- Local anesthetics and procedural medications
- Syringes
- Sterile prep kits and drapes
Nerve Blocks
Chronic Migraine Injection
Member Programs Available (INJX/ASPN)
Built for High-Volum
Procedure Days
Pain management practices perform dozens of procedures every week. Epidural steroid injections, medial branch blocks, and nerve blocks all rely on consistent access to sterile consumables and injectables.
Pipeline Medical helps practices keep procedure-day supplies consistent, available, and ready when needed.
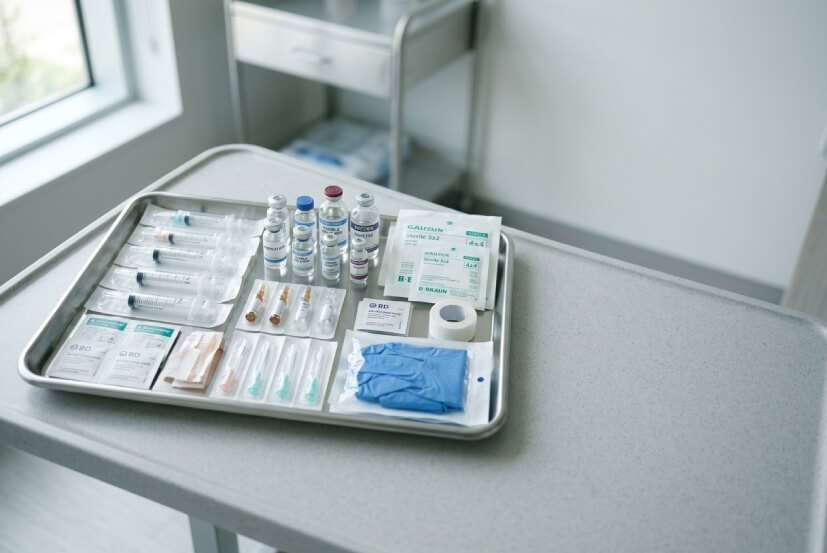
Supporting High-Volume Pain Management Practices
See how pain management teams are streamlining injectables, procedure supplies, and pharmaceutical sourcing with Pipeline Medical.
Pain Management FAQs
Product Access, Fulfillment, and Compliance with Pipeline Medical.
Who can order pain management supplies from Pipeline Medical?
What types of pain management products are available?
Do you offer supplies for image-guided and interventional pain procedures?
Yes. We support practices that perform epidural steroid injections, medial branch blocks, facet injections, nerve blocks, radiofrequency ablation (RFA), and other fluoroscopy- or ultrasound-guided procedures. Our inventory supports high-frequency interventional case schedules.
Do you provide custom procedure trays for pain management practices?
How quickly are pain management orders fulfilled?
Can I view pricing before approval?
Compliance & Quality
Credential Verification: All prescription products are Rx Only and intended for purchase and use by licensed healthcare professionals.
Sterility & Handling: All sterile products are packaged and handled according to the manufacturer’s Instructions for Use.
Botulinum Toxin Products: These carry an FDA Boxed Warning regarding the risk of distant spread of the toxin’s effects. Always review the full Prescribing Information and Medication Guide before ordering or administering.
Safe Use: All products should be used strictly in accordance with the manufacturer’s Instructions for Use (IFU), including proper storage, handling, and application guidelines.
Authorized Sourcing: Pipeline Medical sources pharmaceuticals and interventional supplies exclusively through authorized U.S. manufacturers and distributors with full lot traceability.
Trademarks: All product names and trademarks remain the property of their respective owners.
Built for High-Frequency Interventional Pain Practices That Cannot Afford Supply Disruption
Keep injectables and interventional supplies aligned with your procedure schedule through one secure, credentialed platform.
*Pricing becomes visible after credential verification.
Specialties
Company
© Copyright 2025 | Pipeline Medical, LLC. All rights reserved.
Sitemap